PPE Shortage Catapults Human Solutions Tech into Production
The organization was intended to solve unmet needs in the local community. Its founder didn't anticipate that its first project would be face shields.

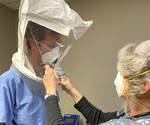
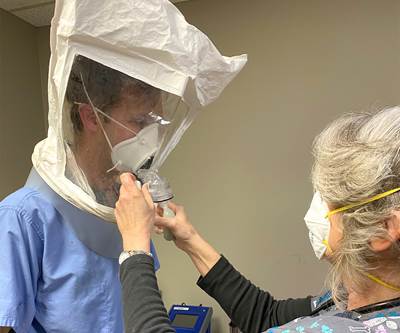
Glen Dobbs is the founder of LoganTech and Human Solutions Tech (HST), a manufacturing organization that will be a nonprofit serving unmet needs in the Connecticut area. Necessity brought about HST’s first project: face shields to protect healthcare professionals from COVID-19.
Human Solutions Tech (HST) was never meant to launch in a pandemic. In fact, just two months ago founder Glen Dobbs expected the coronavirus to delay his work on this nonprofit. Now, the organization is closing in on 500 units shipped of its first product: face shields.
Dobbs is the president and CEO of LoganTech, a Connecticut maker of communications devices for verbally and visually impaired individuals, named for his son Logan. LoganTech develops and manufactures devices that measurably improve the lives of users and their families, but Dobbs has also seen other needs go unmet, needs that this company is not necessarily flexible or agile enough to fill. He conceived of Human Solutions Tech as a separate nonprofit manufacturing organization that would have the freedom to make items such as mobility devices, prosthetics and human/machine interfaces without disrupting operations at LoganTech.
Personal protective equipment (PPE) wasn’t necessarily part of the plan, but when Dobbs learned of the shortage and began networking with local medical facilities he saw the same kind of gap. “PPE is a great example of an unmet need that we envisioned finding a way to meet when we first started discussing creating HST,” Dobbs says.
“3D printing may not always be the right process, but it can usually be the ‘right now’ process.”
He and his staff zeroed in on face shields, and quickly developed a design that combines a laser-cut PETG shield with FDM 3D printed fasteners and elastic hooks. “3D printing has been critical to this project as it has allowed for rapid design iterations,” Dobbs says. “We are setting our sights on having the most agile and flexible set of tools at our disposal, to allow us to rapidly achieve our mission. To borrow from an old saying, 3D printing may not always be the right process, but it can usually be the ‘right now’ process.”
The combination of rapid design, 3D printing and laser cutting technology allowed Human Tech Solution to respond quickly while larger manufacturers ramped up. The organization has delivered almost 500 face shields to date, primarily to healthcare sites in Connecticut.

3D printing isn’t always the right solution, but it can often be the fastest one. HST is delivering face shields made with these printed fasteners often while healthcare facilities wait for PPE to arrive from larger manufacturers.
This fast, local manufacturing model is one that has been echoed throughout the coronavirus period, and one that 3D printing has proven particularly well-suited to support. The digital process, with no need for tooling, has enabled rapid product development for PPE, test swabs and more, and has allowed manufacturers to quickly pivot (or launch, in this case) into making the production items needed most.
Once the demand for face shields subsides, HST will go forward with seeking nonprofit status and advancing its mission. Dobbs envisions a future where other manufacturers donate their time, equipment and expertise to the organization to help continue filling specific, local needs. The face shields it is making today for healthcare professionals are just the first step.
“To be clear, we are not aspiring towards production of PPE — there are plenty of other organizations more well positioned to take this on,” Dobbs says. “We are simply attempting to address local unmet needs that we can respond to right here and now. We absolutely don’t want to set limits on what we expect to be able to do in the future. ”
Related Content
3D Printing with Plastic Pellets – What You Need to Know
A few 3D printers today are capable of working directly with resin pellets for feedstock. That brings extreme flexibility in material options, but also requires greater knowledge of how to best process any given resin. Here’s how FGF machine maker JuggerBot 3D addresses both the printing technology and the process know-how.
Read MoreHow to Improve Polymer AM Productivity 20X
A fast cycle time is critical to efficient production 3D printing, but it’s not the only thing. How you choose the right parts for AM, prepare jobs for production, and manage post processing will have just as big an impact on total 3D printing throughput. It all needs to work together to achieve maximum productivity.
Read MoreWhat Does Additive Manufacturing Readiness Look Like?
The promise of distributed manufacturing is alluring, but to get there AM first needs to master scale production. GKN Additive’s Michigan facility illustrates what the journey might look like.
Read MorePossibilities From Electroplating 3D Printed Plastic Parts
Adding layers of nickel or copper to 3D printed polymer can impart desired properties such as electrical conductivity, EMI shielding, abrasion resistance and improved strength — approaching and even exceeding 3D printed metal, according to RePliForm.
Read MoreRead Next
What the Coronavirus Crisis Will Mean for Additive Manufacturing
In the response to COVID-19, 3D printing demonstrated some of its strengths, but not all of them. However, the full effect of this crisis for AM is still to come.
Read More3D Printing and Coronavirus: U.S. Additive Manufacturers Share Their Experiences
The COVID-19 outbreak has brought both setbacks and opportunities for American manufacturing. 3D printing companies share their stories.
Read MoreAerospace Manufacturer Pivots to 3D Print Masks for COVID-19 First Responders
Rapid Application Group is finalizing a 3D printed mask to protect first responders and medical staff who may be exposed to coronavirus. The nylon mask uses a replaceable N-95 or HEPA filter.
Read More
.jpg;width=70;height=70;mode=crop)